
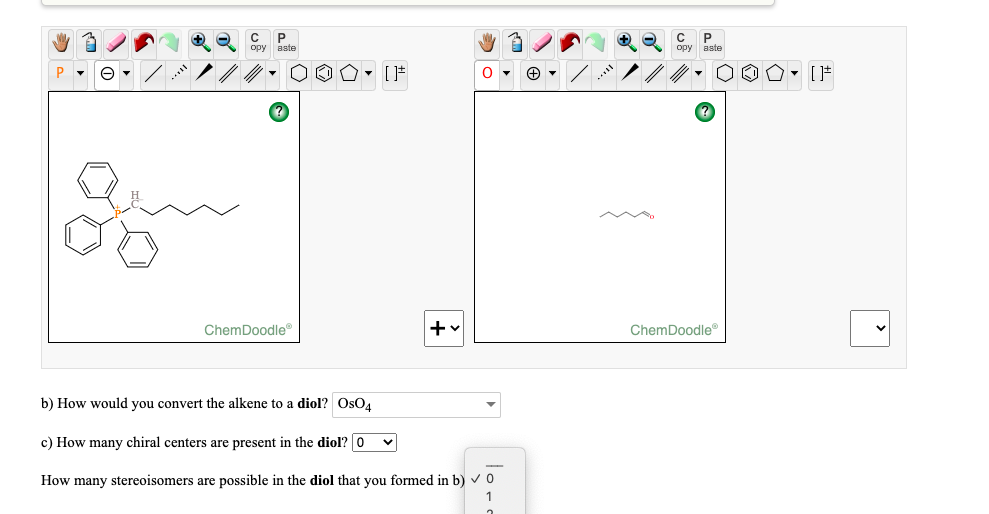
mmod), Schrödinger Maestro (.mae), Standard Molecular Data (.smd), Tripos Mol2 (.mol2. rd), MDL RXNFiles, both V2000 and V3000 connection tables (.rxn), MMI SketchEl Molecule (.el), Molinspiration JME String (.jme), RCSB MacroMolecular Transmission Format (.mmtf), RCSB Protein Data Bank Files (.pdb. dx), ISIS Sketch File (.skc), ISIS Sketch Transportable Graphics File (.tgf), MDL MOLFiles, both V2000 and V3000 connection tables (.mol. smiles), IUPAC InChI (.inchi), IUPAC JCAMP-DX (.jdx. (b) Convergent vs Linear synthesis When considering a disconnection in the retrosynthetic analysis of a complex target molecule, try (if possible) to divide the molecule into halves at convenient bonds. help us caption & translate this video! v c0mb0 a video covering the core mechanistic and stereochemical aspects of the wittig reaction for the second year module organic and biological chemistry.Read and write many popular chemical file types for working with the applications you use:ĪCD/ChemSketch Documents (.sk2), ChemDoodle Documents (.icl), ChemDoodle 3D Scenes (.ic3), ChemDoodle Javascript Data (.cwc.js), CambridgeSoft ChemDraw Exchange (.cdx), CambridgeSoft ChemDraw XML (.cdxml), Crystallographic Information Format (.cif), CHARMM CARD File (.crd), ChemAxon Marvin Document (.mrv), Chemical Markup Language (.cml), Daylight SMILES (.smi. an optimal synthetic route for which reagents are readily available and inexpensive. in my channel you can get chemistry tricks that helps you to attempt your exams without stress.thanks for watching donate here: aklectures donate website video link: aklectures lecture the wittig reaction facebook link: ethyl trans cinnamate is synthesized via the wittig reaction. if you want all in this video i shared wittig reaction. i also provide some hey guys, have you ever heard of the wittig reaction? it's a really really cool and useful reaction that you all will learn in organic chemistry ii that involves chad breaks down the wittig reaction including the formation of a phosphoylide and its addition to a ketone or aldehyde to produce an alkene. triphenylphosphine? ylides? can this get any cooler? let's use wittig reactions to make joechem.io videos 38 for video on joechem and attached worksheet solution (below video on joechem aka the link) in this video, we talk about the it's time for the wittig reaction mechanism to be made easy! follow me in my walk through video and we can do the mechanism together. A phosphonium ylide can be prepared rather straightforwardly. The positive charge in these Wittig reagents is carried by a phosphorus atom with three phenyl substituents and a bond to a carbanion.Ylides can be stabilised or non-stabilised.
subscribe: don't say it like wittig, say it like vittig. Phosphonium ylides are used in the Wittig reaction, a method used to convert ketones and especially aldehydes to alkenes. This organic chemistry video tutorial provides a basic introduction into the wittig reaction mechanism.

the wittig reaction was discovered in 1954 by georg wittig, for which he was awarded the nobel prize in chemistry in 1979. The wittig reaction or wittig olefination is a chemical reaction of an aldehyde or ketone with a triphenyl phosphonium ylide (often called a wittig reagent) to give an alkene and triphenylphosphine oxide. it uses a carbonyl compound as an electrophile, which is attacked by a “phosphorus ylide” (the “wittig reagent”.) while many other routes to alkenes can proceed via elimination. Wittig reaction 53 the wittig reaction: synthesis of alkenes intro the “wittig reaction” is one of the premier methods for the synthesis of alkenes. if a potassium base was used to generate the phosphine oxide anion, the reaction with a carbonyl compound proceeded as in. this modification allows for the removal of phosphorous as a water soluble side product. Horner wittig olefination in 1958, horner and co workers described the use of phosphine oxides in wittig type reactions. if r is an electron withdrawing group, then the ylide is stabilized and is not as reactive as when r is alkyl. the geometry of the resulting alkene depends on the reactivity of the ylide. The wittig reaction allows the preparation of an alkene by the reaction of an aldehyde or ketone with the ylide generated from a phosphonium salt.
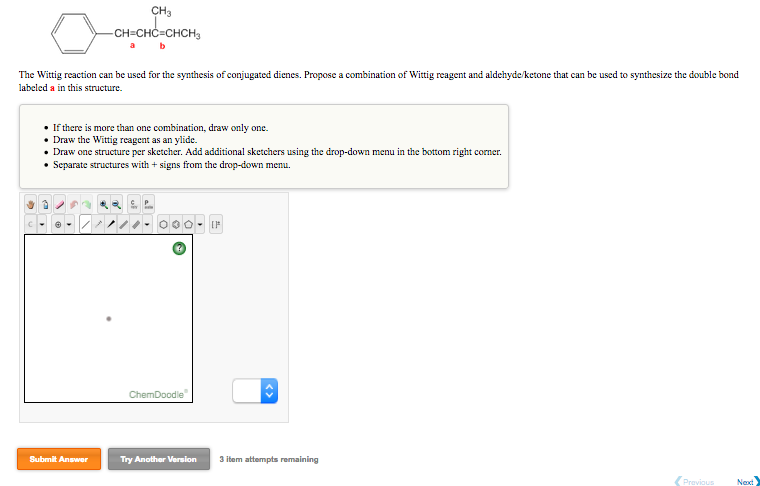
ylides react to give substituted alkenes in a transformation called the wittig reaction. Problem number 61 Fromthe Smith Organic chemistry textbook. the most important use of ylides in synthesis comes from their reactions with aldehydes and ketones, which are initiated in every case by a covalent bonding of the nucleophilic alpha carbon to the electrophilic carbonyl carbon.
